What Does the Second Quantum Number L Describe
It refers to what energy level it is and will be one greater than the. It is the principle quantum number.
The Physical Significance Of The 4 Quantum Numbers
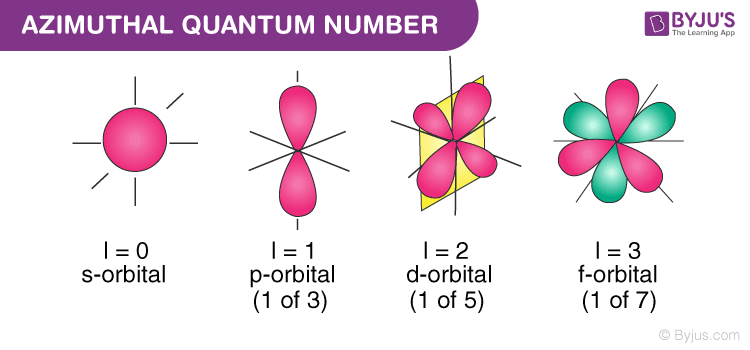
The angular quantum number l describes the shape of the orbital.

. The second quantum number or l which describes the subshell s p d f etc. This means that the electrons in that energy level are only found in the s orbital as that corresponds. The value is 1 or higher never 0 or negative.
It is the principle quantum number. It is the principle quantumnumber. What does the fourth quantum number describe.
Want this question answered. The second quantum number is often called the azimuthal quantum number l. The azimuthal quantum number ℓ is the quantum number associated with the angular momentum of an atomic electron.
The principal quantum number is an integer that is the number of the electrons shell. Shape of electron cloud. The electrons in the first energy level have an angular quantum number l0.
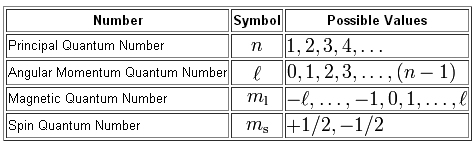
Each value of n has multiple values of ℓ ranging in values from 0 to n-1This quantum number determines the. Electronic quantum numbers quantum numbers that describe electrons are numerical quantities that provide solutions to the Schrodinger wave equation for hydrogen atoms. What does the second quantum number l describe.
L is the secondquantum number. The angular momentum quantum number l describes the shape of the subshell and its orbitals where l 0123. What spin a specific electron has B.
Which sublevel the electron is in O C. The angular momentum quantum number. The azimuthal or orbital angular momentum quantum number describes the shape of a given orbital.
Electron position within the sublevel. It refers to what energy level it is and will be one greater than the. Principal energy level of the electron.
What does the second quantum number l describe. What do quantum numbers describe. N is the first quantum number.
What does the second quantum number 1 describe. What does the third quantum number describe. The Azimuthal Quantum Number.
Be notified when an answer is posted. They can even take on. The second quantum number is the angular quantum number ℓ.
Which energy level is being occupied O D. It refers to what energy level it is and will be onegreater than the number of nodes in the orbital. The second quantum number known as the angular or orbital quantum number describes the subshell and gives the magnitude of the orbital angular momentum through the relation.
The second quantum number known as the angular or orbital quantum number describes the subshell and gives the magnitude of the orbital angular momentum through the. It is also known as the angular momentum quantum. The value of l describes the shape of the region of space.
Quantum numbers are a set of variables which describe pertinent characteristics of electrons within an atom. The azimuthal quantum number also known as the angular momentum quantum number or orbital quantum number describes the subshell and gives the magnitude of the orbital. What is the second quantum number of a 3p3 electron in phosphorus 1s22s22p63s23p3.
What does the second quantum number describe. N is the first quantum number. It is denoted by the symbol l and its value is equal to the total number of angular nodes.
What do quantum numbers describe. Orbitals have shapes that are best described as spherical l 0 polar l 1 or cloverleaf l 2. Tell what the 4 quantum numbers represent.
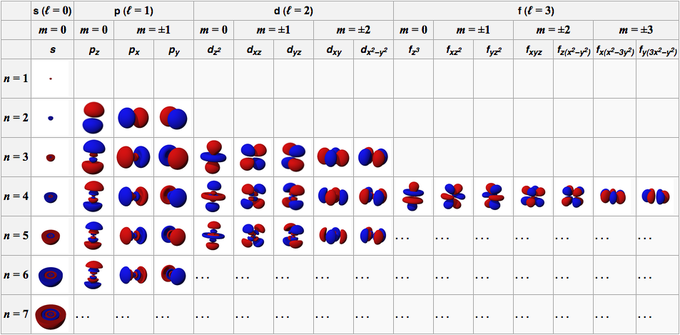
Corresponds to spd and f subshells containing spdf.
Azimuthal Quantum Number Definition Subsidiary And Angular Momentum Quantum Numbers
Quantum Numbers Introduction To Chemistry

Comments
Post a Comment